Experts say scientific understanding of deep hydrocarbons has been transformed, with new insights gained into the sources of energy that could have catalyzed and nurtured Earth’s earliest forms of life.
During the past hundred years scientists worked out in detail how hydrocarbons—fossil fuels” drawn from reservoirs in Earth’s crust to heat and power homes, vehicles, and industry—have a biotic origin, derived from the buried plants, animals, and algae of eons past.
But for some hydrocarbons, especially methane—the colorless, odorless main ingredient in natural gas—nature has many recipes, some of which are “abiotic—derived not from the decay of prehistoric life, but created inorganically by geological and chemical processes deep within the Earth.
Abiotic hydrocarbons have been a major focus of the Deep Energy community of the Deep Carbon Observatory program—a 10-year exploration of Earth’s innermost secrets, concluding in October.
DCO experts believe an abiotic origin of methane explains most of the unusual occurrences of the gas, including the flames of Chimaera in southwest Turkey.
Chimaera does not sit atop conventional deposits of oil and gas produced from the decayed organic residue of earlier epochs. And yet, dozens of small fires have burned at this mountaintop site for millennia.
Ancient explanations for the flames included the breath of a monster—part lion, part goat, part snake. The less colorful scientific reason: highly flammable abiotic methane and hydrogen rising to Earth’s surface from deep below.
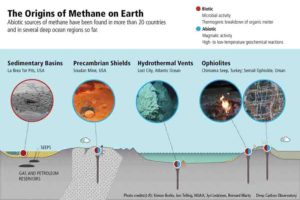
Chimaera is among the most photogenic and famed of now hundreds of sites where abiotic sources of methane have been found in more than 20 countries and in several deep ocean regions so far.
DCO collaborator Giuseppe Etiope of the Istituto Nazionale di Geofisica e Vulcanologia in Rome has documented the Chimaera site and several other environments at which unusual occurrences of methane have been found, including:
- Ancient Precambrian shields—rock at the core of the continents formed as much as 3 billion years ago
- On the ocean floor (e.g., high-temperature vents on and near mid-ocean ridges and belching mud volcanoes)
- On continents (seeps and hyper-alkaline springs and aquifers).
While diverse rock types are present in all these environments, he notes, many discoveries have focused on places with specific, suitable types of “ultramafic” rocks such as peridotite (a coarse-grained igneous rock) included in massifs and ophiolites (ensembles of rocks formed from the submarine eruption of oceanic crustal and upper mantle material).
Earth’s abiotic methane is now thought mainly to derive chemically from the hydrogen created by the hydration of ultramafic rocks undergoing “serpentinization”—a reaction that occurs when water meets the mineral olivine.
Hydrogen also nourishes biological sources of methane. DCO researchers have documented a vast microbial ecosystem—a deep biosphere fed by hydrogen. Many of the deep microbes, called methanogens, metabolize hydrogen to produce methane.
The deep biosphere has therefore posed a chicken and egg scenario: which came first, abiotic methane or microbes? If abiotic methane came first, as seems obvious, did it give rise to Earth’s first microbes? And if microbes came first, how and why did they inhabit places almost devoid of sustenance?
A decadal goal: sort out the origins of methane on Earth
When the Deep Carbon Observatory project began in 2009, DCO’s Deep Energy community—now made up of more than 230 researchers from 35 nations, set the decadal goal of sorting out the origins of methane on Earth.
Some hypothesized that unusual methane reservoirs—i.e., those that could not be biotic in origin—must form through chemical reactions occurring in the surrounding rocks.
Others suggested that microbes contributed to methane production in some reservoirs, metabolizing hydrogen to create methane in an entirely different process.
Others hypothesized that methane might originate deeper in Earth, in the upper mantle, and diffuse up toward the surface. (At Moscow’s Gubkin University, researcher Vladimir Kutcherov is leading experiments to test the production of methane in lab-simulated high-pressure conditions of Earth’s upper mantle).
Early in its mandate the DCO made the decision to invest in new analytical instrumentation to overcome some of the limitations to deciphering the origin of methane.
With strategic investment in instrumentation and numerous field samples, DCO partners set out to pioneer new investigative tools to distinguish Earth’s biotic from abiotic methane.
In 2014, three new instruments came online with the potential to change the face of deep carbon science, and they have not disappointed, says Edward Young, of the University of California, Los Angeles (UCLA), co-leader of DCO’s Deep Energy Community with Isabelle Daniel of the Claude Bernard University Lyon 1 in Lyon, France.
Using complementary techniques of mass spectrometry and absorption spectroscopy, scientists at UCLA, the California Institute of Technology (Caltech), Pasadena CA, and the Massachusetts Institute of Technology (MIT), Cambridge MA, are analyzing natural methane samples to better understand how abiotic methane may be produced.
“A molecule of methane (CH4) appears remarkably simple, made up of only five atoms,” says Dr. Young. “Rare isotopes of both hydrogen and carbon are occasionally incorporated into methane molecules, however, and the frequency of these ‘heavy’ isotopes reveals the secret of how they formed and at what temperatures.”
Of particular diagnostic value are methane molecules that contain more than one “heavy” isotope (“clumped isotopes”). These molecules are extremely rare and can only be distinguished by instruments with extremely high mass resolution, sensitivity, and power.
DCO collaborators used samples of gases collected from Chimaera, the deep mines of Canada, the Oman ophiolite, hydrothermal vents on the ocean floor, and additional sites, and were surprised by what they found.
Though interpreting the data is challenging, it appears microbes may be doing more than originally thought.
How much abiotic methane?
“We see curious biological fingerprints in samples that otherwise appear to have an abiotic signature,” says Dr. Daniel. “It seems microbes know how to use these abiotic compounds as fuel.”
“We have clear and growing evidence of abiotic methane on Earth. What is not clear is how much there is. These investigations have found incredible complexity in the way methane is produced, and these complexities connect inorganic and organic chemistry on Earth in fascinating ways.”
Adds Dr. Young: “We went into this project thinking we knew how abiotic methane formed. What we’re learning is that it is much more complicated, and the biggest key is hydrogen. With greater understanding of how rocks make the hydrogen from which methane derives, and how fast this reaction happens, we’ll be a lot closer to knowing how much methane there is on Earth.”
Jesse Ausubel of The Rockefeller University in New York notes that the popular definition of “fossil fuel” doesn’t cover abiotic methane.
“Thousands of samples from many settings tested with super-sensitive instruments are producing a global picture of the abundances and fluxes of deep energy. Much of the very deep hydrocarbons is not conventional fossil fuel, as popularly defined.”
The behaviors of biotic and abiotic methane, it should be noted, in terms of energy output and emissions when burned, are indistinguishable.
Key findings to date:
- Thanks to new instruments, scientists have identified new isotope signatures in methane to help determine its provenance—an impossibility 10 years ago
- The serpentinization reaction is better understood and is one of several ways Earth’s rocks produce molecular hydrogen—a key source of geologic energy for the deep biosphere
- That hydrogen reacts with carbon dioxide to produce methane was long known. How this happens in Earth’s crust, however, is highly complex, and many other organic molecules are created as byproducts in the process. These molecules can be used by microbes as a food source. They also represent intriguing clues as to the origins of life on Earth, as these organic molecules may be precursors for the building blocks of life (e.g., amino acids)
- With similar conditions and reactions likely on other planets and moons (e.g., the subsurface of Mars or on the ocean floor of Enceladus), it strengthens the potential identification of where life may exist elsewhere in the universe
- Studies of serpentinizing systems have found other abiotic hydrocarbons in addition to methane.
Future implications:
These investigations into how abiotic methane forms on Earth are not the end of the story, but rather the beginning.
The last 10 years have seen transformational changes in our understanding of the origins of methane on Earth and its pivotal role in sustaining the deep biosphere, providing a glimpse into the geological processes that could have set the stage for life.
With these new discoveries, we are poised to answer numerous big questions, such as:
- How much abiotic methane is being produced in Earth?
- How much methane do the microbes of Earth’s deep biosphere produce?
- How much do the microbes consume?
- What are movements and fates of abiotic methane?and
- Where is abiotic methane stored and for how long?
The success of the project’s research has not only changed perceptions of energy generation in deep Earth, but also about how life may have found a foothold on our planet.
And if abiotic energy does occur on Earth, how likely is it that similar reactions and life have occurred elsewhere in the cosmos?
This Deep Energy research released today is a result of the Deep Carbon Observatory program, which will issue its final report in October 2019 after a decade of work by a global community of more than 1000 scientists to better understand the quantities, movements, forms, and origins of carbon inside Earth.
Reference:
- The contribution of the Precambrian continental lithosphere to global H2 production
Sherwood Lollar, B., Onstott, T.C., Lacrampe-Couloume, G., and Ballentine, C.J. (2014). Nature 516 (7531): 379-382. - Formation temperatures of thermogenic and biogenic methane
Stolper DA, Lawson M, Davis CL, Ferreira AA, Santos Neto EV, Ellis GS, Lewan MD, Martini AM, Tang Y, Schoell M, Sessions AL, Eiler JM (2014). Science 344:1500-1503 - Measurement of a doubly-substituted methane isotopologue, 13CH3D, by tunable infrared laser direct absorption spectroscopy
Ono S, Wang DT, Gruen DS, Sherwood Lollar B, Zahniser M, McManus BJ, Nelson DD (2014), Analytical Chemistry, 86:6487-6494 - Panorama, a new gas source, electron impact, double-focusing, multi-collector mass spectrometer for the measurement of isotopologues in geochemistry
Young ED, Freedman P, Rumble D, Schauble E (2014), 7th International Symposium on Isotopomers (ISI2014), Tokyo, Japan - The relative abundances of resolved 12CH2D2 and 13CH3D and mechanisms controlling isotopic bond ordering in abiotic and biotic methane gases
Young E.D., Kohl I.E., Sherwood Lollar B., Etiope G., Rumble III D., Li S., Haghnegahdar - M.A., Schauble E.A., McCain K.A., Foustoukos D.I., Sutclife C., Warr O., Ballentine C.J., Onstott T.C., Hosgormez H., Neubeck A., Marques J.M., Pérez-Rodríguez I., Rowe A.R., LaRowe D.E., Magnabosco C., Yeung L.Y., Ash J.L., Bryndzia L.T. (2017). Geoch. Cosmochim. Acta, 203, 235-264.
- Natural gas seepage, the Earth’s Hydrocarbon Degassing
G. Etiope. (2015), Springer, Switzerland - Widespread abiotic methane in chromitites
Etiope G., Ifandi E., Nazzari M., Procesi M., Tsikouras B., Ventura G., Steele A., Tardini R., Szatmari P. (2018). Scientific Reports, 8, 8728, DOI: 10.1038/s41598-018-27082-0. - Massive production of abiotic methane during subduction evidenced in metamorphosed ophicarbonates from the Italian Alps
Vitale Brovarone A, Martinez I, Elmaleh A, Compagnoni R, Chaduteau C, Ferraris C, Esteve I (2017). Nature Communications 8:14134 DOI: 10.1038/ncomms14134
Abiotic formation of condensed carbonaceous matter in the hydrating oceanic crust - Sforna MC, Brunelli D, Pisapia C, Pasini V, Malferrari D, Ménez B. (2018). Nature Communications DOI: 10.1038/s41467-018-07385-6
- Abiotic synthesis of amino acids in the recesses of the oceanic lithosphere
Ménez B, Pisapia C, Andreani M, Jamme F, Vanbellingen QP, Brunelle A, Richard L, Dumas P, Réfrégiers M. (2018). Nature DOI: 10.1038/s41586-018-0684-z
Abiotic methane on Earth
Etiope G, Sherwood Lollar B (2013). Reviews of Geophysics DOI: 10.1002/rog.20011 - Formation of abiotic hydrocarbon from reduction of carbonate in subduction zones: Constraints from petrological observation and experimental simulation
Tao R, Zhang L, Tian M, Zhu J, Liu X, Liu J, Höfer HE, Stagno V, Fei Y (2018) Geochimica et Cosmochimica Acta 239:390 DOI: 10.1016/j.gca.2018.08.008 - Immiscible hydrocarbon fluids in the deep carbon cycle
Huang, F., Daniel I., Cardon H., Montagnac G., Sverjensky D. (2017) Nature Communications 8:15798 DOI: 10.1038/ncomms1579 - Methane-derived hydrocarbons produced under upper-mantle conditions
A. Kolesnikov, V.G. Kutcherov, A.F. Goncharov (2009) Nature Geoscience 2: 566-570 - Synthesis of Complex Hydrocarbon Systems at Temperatures and Pressures Corresponding to the Earth’s Upper Mantle Conditions
V.G. Kutcherov, A. Kolesnikov, T.I. Dyugheva, L.F. Kulikova, N.N. Nikolaev, O.A. Sazanova, V.V. Braghkin (2010). Doklady Physical Chemistry 433:132-135
Note: The above post is reprinted from materials provided by Deep Carbon Observatory .