A thorough examination of tiny crystals of zircon, a mineral found in rhyolites, an igneous rock, from the Snake River Plain has solidified evidence for a new way of looking at the life cycle of super-volcanic eruptions in the long track of the Yellowstone hotspot, say University of Oregon scientists.
The pattern emerging from new and previous research completed in the last five years under a National Science Foundation career award, said UO geologist Ilya N. Bindeman, is that another super-eruption from the still-alive Yellowstone volcanic field is less likely for the next few million years than previously thought (see related story, “Not in a million years, says Oregon geologist about Yellowstone eruption”). The last eruption 640,000 years ago created the Yellowstone Caldera and the Lava Creek Tuff in what is now Yellowstone National Park.
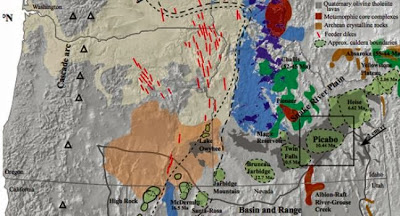
The Yellowstone hotspot creates a conveyor belt style of volcanism because of the southwest migration of the North American plate at 2-4 centimeters (about .8 to 1.6 inches) annually over the last 16 million years of volcanism. Due to the movement of the North American plate, the plume interaction with the crust leaves footprints in the form of caldera clusters, in what is now the Snake River Plain, Bindeman said.
The Picabo volcanic field of southern Idaho, described in a new paper by a six-member team, was active between 10.4 and 6.6 million years ago and experienced at least three, and maybe as many as six, violent caldera-forming eruptions. The field has been difficult to assess, said lead author Dana Drew, a UO graduate student, because the calderas have been buried by as much as two kilometers of basalt since its eruption cycle died.
The work at Picabo is detailed in a paper online ahead of publication in the journal Earth and Planetary Science Letters.
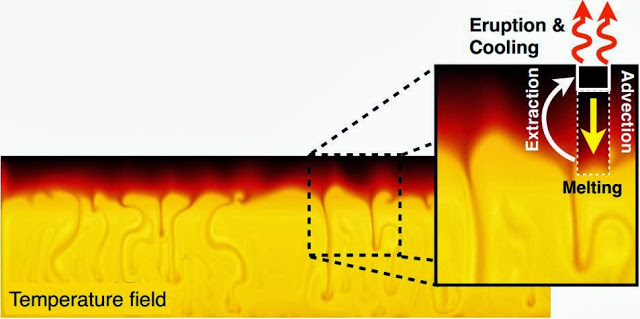
The team theorized that basalt from the mantle plume, rocks from Earth’s crust and previously erupted volcanoes are melted together to form the rhyolites erupted in the Snake River Plain. Before each eruption, rhyolite magma is stored in dispersed pockets throughout the upper crust, which are later mixed together, according to geochemical evidence. “We think that this batch-assembly process is an important part of caldera-forming eruptions, and generating rhyolites in general,” Drew said.
In reaching their conclusions, Drew and colleagues analyzed radiogenic and stable isotopic data — specifically oxygen and hafnium — in zircons detected in rhyolites found at the margins of the Picabo field and from a deep borehole. That data, in combination with whole rock geochemistry and zircon uranium-lead geochronology helped provide a framework to understand the region’s ancient volcanic past.
Previous research on the related Heise volcanic field east of Picabo yielded similar results. “There is a growing database of the geochemistry of rhyolites in the Yellowstone hotspot track,” Drew said. “Adding Picabo provides a missing link in the database.
Drew and colleagues, through their oxygen isotope analyses, identified a wide diversity of oxygen ratios occurring in erupted zircons near the end of the Picabo volcanic cycle. Such oxygen ratios are referred to as delta-O-18 signatures based on oxygen 18 levels relative to seawater. (Oxygen 18 contains eight protons and 10 neutrons; Oxygen 16, with eight protons and eight neutrons, is the most commonly found form of oxygen in nature.)
The approach provided a glimpse into the connection of surface and subsurface processes at a caldera cluster. The interaction of erupted rhyolite with groundwater and surface water causes hydrothermal alteration and the change in oxygen isotopes, thereby providing a fingerprinting tool for the level of hydrothermal alteration, Drew said.
“Through the eruptive sequence, we begin to generate lower delta-O-18 signatures of the magmas and, with that, we also see a more diverse signature,” Drew said. “By the time of the final eruption there is up to five per mil diversity in the signature recorded in the zircons.” The team attributes these signatures to the mixing of diverse magma batches dispersed in the upper crust, which were formed by melting variably hydrothermally altered rocks — thus diverse delta-O-18 — after repeated formation of calderas and regional extension or stretching of the crust.
When the pockets of melt are rapidly assembled, the process could be the trigger for caldera forming eruptions, Bindeman said. “That leads to a homogenized magma, but in a way that preserves these zircons of different signatures from the individual pockets of melt,” he said. This research, he added, highlights the importance of using new micro-analytical isotopic techniques to relate geochemistry at the crystal-scale to processes occurring at the crustal-wide scale in generating and predicting large-volume rhyolitic eruptions.
“This important research by Dr. Bindeman and his team demonstrates the enormous impact an NSF CAREER award can have,” said Kimberly Andrews Espy, vice president for research and innovation and dean of the graduate school at the University of Oregon. “The five-year project is providing new insights into the eruption cycles of the Yellowstone hotspot and helping scientists to better predict future volcanic activity.”
Note : The above story is based on materials provided by University of Oregon.