Researchers are resurrecting ancient bacterial protein complexes to determine how 3.5-billion-year-old cells functioned versus cells of today. Surprisingly, they are not that different, reports a study published June 9 in Cell Chemical Biology. Despite a popular hypothesis that primordial organisms had simple enzyme proteins, evidence suggests that bacteria around 500 million years after life began already had the sophisticated cellular machinery that exists today.
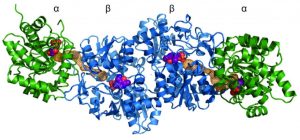
Fossils of 3.5-billion-year-old bacteria are not available, but scientists can reconstruct what their enzymes may have looked like based on phylogenetic trees of proteins from living bacteria. Comparing the amino acid sequences of more than 50 bacteria helped to computationally generate the sequences for the protein subunits of an enzyme complex that was very likely similar to that found in the bacteria’s last common ancestor. The researchers then produced this ancient enzyme complex to study its structure and function.
“There is a generally accepted theory (see bottom citation below) that states that very old enzymes were not as sophisticated as they are now,” says senior author Reinhard Sterner of the Institute of Biophysics and Physical Biochemistry at the University of Regensburg in Germany. “But we used the method of ancestral sequence reconstruction to go back as far as possible in evolutionary time to show that the tryptophan synthase complex from the last bacterial common ancestor was sophisticated–characterized by the high enzymatic activity and communication between subunits seen in modern enzyme complexes.”
“Our data and similar results that have been found by other people suggest that enzymes were already sophisticated 3.5 billion years ago, but this was a surprise because biological evolution started only about 4 billion years ago,” says co-author Rainer Merkl, also at Regensburg. “We conclude that in this very early phase of biological evolution–between 4 billion and 3.5 billion years ago–we probably have primitive enzymes with low efficiency, but this 500 million years was enough time for these enzymes to become fully sophisticated.”
What happened in that 500-million-year gap to place the evolutionary pressures on bacteria to make enzyme complexes is a mystery. Creating these structures is very difficult, as a complex involves multiple subunits that catalyze different reactions in isolation as well as in response to one another. Once formed, however, these complexes have not been seriously altered in billions of years of subsequent evolution, proving their efficiency.
Going forward, Sterner and his colleagues want to continue using the ancestral sequence reconstruction method to better understand the exact steps that led to the formation of the tryptophan synthase complex and its adaptation to specific habitats.
References:
- Busch et al. Ancestral Tryptophan Synthase Reveals Functional Sophistication of Primordial Enzyme Complexes. Cell Chemical Biology, 2016 DOI: 10.1016/j.chembiol.2016.05.009
- R A Jensen. Enzyme Recruitment in Evolution of New Function. Annual Review of Microbiology, 1976; 30 (1): 409 DOI: 10.1146/annurev.mi.30.100176.002205
Note: The above post is reprinted from materials provided by Cell Press.