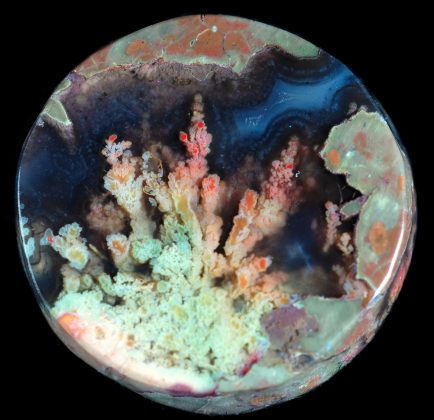
A Thunder Egg “Thunderegg” is a nodule-like rock, similar to a filled geode, that is formed within rhyolitic volcanic ash layers. Thundereggs are rough spheres, most about the size of a baseball—though they can range from less than an inch to over a meter across. They usually contain centres of chalcedony which may have been fractured followed by deposition of agate, jasper or opal, either uniquely or in combination. Also frequently encountered are quartz and gypsum crystals, as well as various other mineral growths and inclusions. Thundereggs usually look like ordinary rocks on the outside, but slicing them in half and polishing them may reveal intricate patterns and colours. A characteristic feature of thundereggs is that (like other agates) the individual beds they come from can vary in appearance, though they can maintain a certain specific identity within them.
Thunder Egg is not synonymous with either geode or agate. A geode is a simple term for a rock with a hollow in it, often with crystal formation/growth. A thunderegg on the other hand is a specific geological structure. A thunderegg may be referred to as a geode if it has a hollow in it (see illustration of Gehlberg specimen), but not all geodes are thundereggs because there are many different ways for a hollow to form. Similarly, a thunderegg is just one of the forms that agate can assume.
Many thundereggs found at Rockhound State Park are spherical and consist of two distinct parts: a dark-gray to pinkish outer part and a white, blue, or gray inner part, or core, which is recognizable as agate, chalcedony, and quartz crystals, all forms of the compound SiO2. In many examples, these two parts can be described as a shell and a filling. However, some thundereggs, or spherulites, do not contain the filling; they are composed of solid dark-gray to pinkish shell material (Fig. 3) or are partly hollow. Geologically distinct processes form the two parts of the thundereggs. The outer part of the thundereggs is formed by complex magmatic processes (i.e. as spherulites), and then the inner part is formed and modified by multiple cycles of late-stage hydrothermal fluids. The processes that form geodes and thundereggs are complex and are controlled by constantly changing physical and chemical conditions, such as temperature, pressure, depth of formation, composition of the magma, composition of the ground water, and composition of the host rocks.
Occurrence
Thundereggs are found globally wherever conditions are right. In the USA, Oregon remains one of the most famous thunderegg locations. Germany is also an important center for thunderegg agates (especially sites like St Egidien and Gehlberg). Other countries known for their thundereggs include some places in Africa, Poland, Romania, Turkey, Mexico, Argentina, Canada, Australia and France.
Formation
Thundereggs are found in flows of rhyolite lava. They form in gas pockets in the lava, which act as molds, from the action of water percolating through the porous rock carrying silica in solution. The cooled bubbles were gradually filled by water percolating through the porous rock carrying rich quantities of silica (quartz). The deposits lined and filled the cavity, first with a darker matrix material, then an inner core of agate or chalcedony. The various colors come from differences in the minerals found in the soil and rock that the water has moved through.
The agate, chalcedony, and quartz veins and open-space-fillings within voids in the spherulites formed later by multiple cycles of hydrothermal fluids. Hydrothermal fluids are a mixture of late-stage fluids escaping the magma and local ground water. The fluids contain some elements from the original magma and also dissolved minerals from the country rock. The amount of ions that the fluid can dissolve depends upon pH, temperature, pressure, and composition of the fluid. The hydrothermal fluids move through fractures in the rocks, which crosscut the igneous textures, and form veins or banded agate, chalcedony, and quartz. Some of these fluids seep through microscopic pores and into spherulites and gas pockets in the volcanic rocks, and they precipitate crystals along the walls of the cavity, forming geodes and geode-like spherulites. Other fluids seep through fractures and gas and other void spaces in the spherulites. Because of their formation by multiple hydrothermal events, each thunderegg provides clues as to its unique formation.
Different temperatures and fluid compositions would account for the variety of textures found within any given thunderegg or geode. By carefully studying the crystal fill and textures in spherulites and geodes, geologists can piece together the different processes through time that formed them. The banding found within some spherulites and geodes consists of multiple layers of different colored agate, chalcedony, and locally quartz, and may have been formed by fluids supersaturated in silica (Fournier, 1985a). Supersaturated solutions are solutions that contain excessive silica in solution. The presence of silica minerals within the thundereggs and geodes indicates that the fluids were saturated in silica. Saturated fluids are fluids that contain enough silica in solution without precipitating silica minerals. When a silica-saturated solution cools slowly, crystalline quartz is deposited at approximately 200°–340° C (Fournier, 1985a). Rapid cooling of a silica-saturated fluid allows supersaturated solutions to form that precipitate chalcedony or amorphous silica. These supersaturated fluids are unstable and quickly deposit thin layers of chalcedony or amorphous silica, typically at lower temperatures (<200° C). The fluid loses silica due to precipitation and becomes saturated with silica but as the fluid continues to cool rapidly, it becomes supersaturated with silica again. An increase in salinity (such as NaCl) increases the solubility of silica at higher temperatures and also produces saturated silica solutions (Fournier, 1985a). Supersaturation of the fluids can also occur by mixing of different hydrothermal fluids, especially with different pHs, and by reaction of hydrothermal fluids with volcanic gases.
The different colors of the bands are a result of trace amounts of impurities, such as iron (red), manganese (black, pink), cobalt (blue, violet-red), copper (green, blue), chromium (orange-red), nickel (green), etc. Faceted quartz crystals indicate that the fluids were somewhat supersaturated with silica and that precipitation occurred under relatively slow-changing conditions (Fournier, 1985a).
Not all geodes are spherulites formed by magmatic processes; other natural processes form some geodes. Lower-temperature ground water percolates through the cooled volcanic rocks and dissolves additional minerals. These fluids are typically low temperature (<200° C), although locally higher-temperature (200°– 300° C) ground waters may be present, especially adjacent to the volcanic vents. These fluids move through microscopic pores in the rock by a process called “diffusion.” During diffusion, some ions in the fluid collect into void spaces and gas pockets in the rock, whereas other ions cannot pass through. This collection of ions surrounding these void spaces may actually, in some cases, form the hard outer shell that is characteristic of geodes. The outer shell may be strengthened by the precipitation of some ions that were excluded during crystallization of agate, chalcedony, and quartz and concentrated in the remaining fluid. Fluids also enter the void spaces through fractures. The void spaces may have originally formed by gas pockets within the magma or by prior dissolution of spherulites or other features within the volcanic rock.
How do the Thunder Egg/spherulites become hollow?
this is difficult to demonstrate directly, we speculate that the hollow centers of spherulites are formed by nucleation, coalescence, and expansion of vapor bubbles at high temperature, resulting in a hollow center that can be filled later by silica. The vapor bubbles would have formed as a result of crystallization of quartz, feldspar, and magnetite, which contain no water, from rhyolitic magma, which contains a small amount of dissolved water at atmospheric pressure (Taylor, 1986). Calculations of the volume of water vapor that could have formed from anhydrous crystallization suggest that the volume would be more than enough to generate the size of hollow cavities seen in spherulites. The reason that some spherulites are hollow and others are not may be related to the rate and depth of crystallization and to the resultant ability or inability of vapor bubbles to nucleate and coalesce.
Reference:
Wikipedia: Thunderegg
Oregon State Rock:State Symbols USA
The Formation of Thundereggs: Robert Paul Colburn PDF
Rock Hound State Park and Spring Canyon Recreation Area, Recreation Area: New Mexico Geology, v. 22, no. 3, p. 66-71, 86.