Algae may hold the key to feeding the world’s burgeoning population. Don’t worry; no one is going to make you eat them. But because they are more efficient than most plants at taking in carbon dioxide from the air, algae could transform agriculture. If their efficiency could be transferred to crops, we could grow more food in less time using less water and less nitrogen fertilizer.
New work from a team led by Carnegie’s Martin Jonikas published in Proceedings of the National Academy of Sciences reveals a protein that is necessary for green algae to achieve such remarkable efficiency. The discovery of this protein is an important first step in harnessing the power of green algae for agriculture.
It all starts with the world’s most abundant enzyme, Rubisco.
Rubisco “fixes” (or converts) atmospheric carbon dioxide into carbon-based sugars, such as glucose and sucrose, in all photosynthetic organisms on the planet. This reaction is central to life on Earth as we know it, because nearly all the carbon that makes up living organisms was at some point “fixed” from the atmosphere by this enzyme. The rate of this reaction limits the growth rate of many of our crops, and many scientists think that accelerating this reaction would increase crop yields.
The funny thing about Rubisco is that it first evolved in bacteria about 3 billion years ago, a time when the Earth’s atmosphere had more abundant carbon dioxide compared to today. As photosynthetic bacteria became more and more populous on ancient Earth, they changed our atmosphere’s composition.
“Rubisco functioned very efficiently in the ancient Earth’s carbon dioxide-rich environment,” Jonikas said. “But it eventually sucked most of the CO2 out of the atmosphere, to the point where CO2 is a trace gas today.”
Rubisco is quite literally a victim of its own success. CO2 makes up only about 0.04 percent of molecules in today’s atmosphere. In this low concentration of CO2, Rubisco works extremely slowly, which limits the growth rates of many crops.
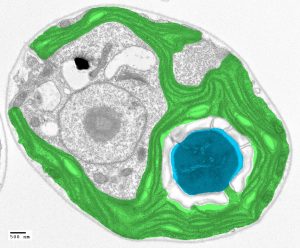
It turns out that algae have evolved a way to make Rubisco run faster. It’s called the pyrenoid. Think of it as a turbocharger for carbon fixation.
The pyrenoid is a tiny compartment inside the cell that is packed with Rubisco and is surrounded by a sheath of starch. Under a microscope, a pyrenoid looks like a spherical bubble inside the cell. Its job is to concentrate carbon dioxide around Rubisco so that Rubisco can run faster.
A pyrenoid provides such a tremendous growth advantage that nearly all algae in the oceans have one. About a third of the planet’s carbon fixation is thought to happen in pyrenoids, yet we know almost nothing about how these structures are formed at a molecular level. Such a molecular understanding is needed before researchers can attempt to engineer pyrenoids into crops, which is expected to enhance crop yields by as much as 60 percent.
The research team focused on a fundamental decades-old mystery: what causes Rubisco to cluster at the core of the pyrenoid?
Jonikas and his team discovered that in their model alga Chlamydomonas, this clustering of Rubisco is mediated by a protein they called EPYC1 for Essential Pyrenoid Component 1. They found that EPYC1 bound with Rubisco and packaged it into the matrix of proteins that forms the pyrenoid’s interior. What’s more, proteins similar to EPYC1 are found in most pyrenoid-containing algae, and are not found in algae that lack these structures.
“A lot of additional work is needed to fully understand EPYC1 and pyrenoids, but our findings are a first step toward engineering algal carbon-capture efficiency into crops,” Jonikas said.
The research team also included Carnegie’s Luke Mackinder (the lead author), Vivian Chen, Elizabeth Freeman Rosenzweig, Leif Pallesen, Gregory Reeves, and Alan Itakura. The project was a close collaboration with Moritz Meyer, Madeline Mitchell, Oliver Caspari, and Howard Griffiths of the University of Cambridge; Tabea Mettler-Altmann, Frederik Sommer, Timo Mühlhaus, Michael Schroda and Mark Stitt of the Max Planck Institute of Molecular Plant Physiology; Robyn Roth and Ursula Goodenough of Washington University St. Louis; and Stefan Geimer of University of Bayreuth.
Note: The above post is reprinted from materials provided by Carnegie Institution for Science.










